- English
- 日本語
- 한국어
- Deutsch
- Français
- Español
No data
官方服务号
招聘公众号
To support ongoing cell therapy manufacturers and developers, ACROBiosystems has developed a wide array of high-quality cytokines for the in vitro culture of immune cells, stem cells, organoids, and various other cell types. We also offer Premium(Pre-GMP) and GMP grades for our cytokines to better meet the needs of various drug development stages and applications. All the cytokines are manufactured using a similar production process, therefore enabling a seamless transition between grades. As a result, we enable you to seamlessly transition between our products and accelerate your research and development.
| Premium(Pre-GMP) Grade | GMP Grade | |
|---|---|---|
| Application | Research and Development; Preclinical research, seamless transition into clinical phases | Designed to meet clinical phase requirements and bolster your IND application to various regulatory bodies. |
| Quality System | ISO 9001 /ISO 13485 Certified | ISO 9001 /ISO 13485 Certified (Development stage) GMP Quality Management System (Production stage) |
| Production | ISO certified facilities | GMP certified facilities |
| Transient or stable cell lines | Stable cell lines (Full inspection according to USP and ChP) | |
| Animal-origin free materials or BSE/TSE free | Animal-origin free materials or BSE/TSE free | |
| Pharmaceutical-grade materials | Pharmaceutical-grade materials | |
| Strict sterilization filtration | Strict sterilization filtration | |
| Class C+A room with manual aseptic filling (ISO5) | B+A Cleanroom aseptic filling | |
| No additional virus clearance steps | Additional specific virus removal processes | |
| Quality Control | Sterility / Mycoplasma testing | Sterility / Mycoplasma testing |
| Endotoxin control and detection | Endotoxin control and detection | |
| Validated key equipment and analytical instruments | Validated equipment /analytical instruments/analytical methods(Audit trail available) | |
| Residual DNA/HCP testing | Residual DNA/HCP testing | |
| Limited adventitious agent testing | Stricter standards for viral residues alongside animal safety evaluations (select products). | |
| Documentation | Common regulatory support | Comprehensive regulatory support files |
| DMF filing (Few products) | DMF filing (All products) |
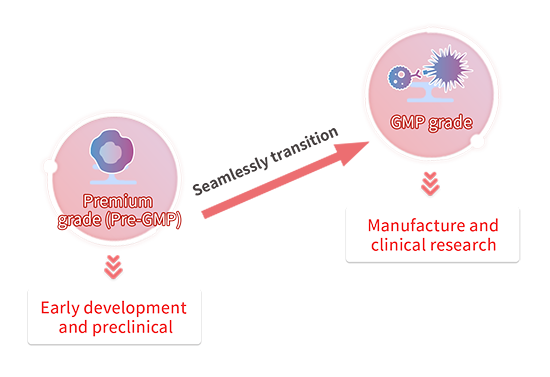
Transitioning from Premium(Pre-GMP) to GMP 
Cytokines are critical raw materials used in cell culturing for cell and gene therapy (CGT) drugs. Usually, in the preclinical stage, both safety and quality for raw materials are not prioritized. In this case, research-use only products can be used. However, when progressing into later drug development stages such as CMC or clinical phases, it is necessary to replace the raw materials with GMP-grade materials to be compliant with regulatory guidelines. In this transitional period, there is a significant amount of energy dedicated to re-evaluating and validating new raw materials.
To ease this transition from the preclinical development into the clinical stage, we offer several grades of cytokines that have been evaluated to be near-identical in bioactivity, along with the documentation required from GMP products. We recommend using our Premium(Pre-GMP) grade raw materials in the early development stage to seamlessly transition to our GMP-grade raw materials when entering CMC or clinical phases and minimize the number of re-evaluation and validation studies performed.
To support ongoing cell therapy manufacturers and developers, ACROBiosystems has developed a wide array of high-quality cytokines for the in vitro culture of immune cells, stem cells, organoids, and various other cell types. We also offer Premium(Pre-GMP) and GMP grades for our cytokines to better meet the needs of various drug development stages and applications. All the cytokines are manufactured using a similar production process, therefore enabling a seamless transition between grades. As a result, we enable you to seamlessly transition between our products and accelerate your research and development.
| Premium(Pre-GMP) Grade | GMP Grade | |
|---|---|---|
| Application | Research and Development; Preclinical research, seamless transition into clinical phases | Designed to meet clinical phase requirements and bolster your IND application to various regulatory bodies. |
| Quality System | ISO 9001 /ISO 13485 Certified | ISO 9001 /ISO 13485 Certified (Development stage) GMP Quality Management System (Production stage) |
| Production | ISO certified facilities | GMP certified facilities |
| Transient or stable cell lines | Stable cell lines (Full inspection according to USP and ChP) | |
| Animal-origin free materials or BSE/TSE free | Animal-origin free materials or BSE/TSE free | |
| Pharmaceutical-grade materials | Pharmaceutical-grade materials | |
| Strict sterilization filtration | Strict sterilization filtration | |
| Class C+A room with manual aseptic filling (ISO5) | B+A Cleanroom aseptic filling | |
| No additional virus clearance steps | Additional specific virus removal processes | |
| Quality Control | Sterility / Mycoplasma testing | Sterility / Mycoplasma testing |
| Endotoxin control and detection | Endotoxin control and detection | |
| Validated key equipment and analytical instruments | Validated equipment /analytical instruments/analytical methods(Audit trail available) | |
| Residual DNA/HCP testing | Residual DNA/HCP testing | |
| Limited adventitious agent testing | Stricter standards for viral residues alongside animal safety evaluations (select products). | |
| Documentation | Common regulatory support | Comprehensive regulatory support files |
| DMF filing (Few products) | DMF filing (All products) |

Transitioning from Premium(Pre-GMP) to GMP 
Cytokines are critical raw materials used in cell culturing for cell and gene therapy (CGT) drugs. Usually, in the preclinical stage, both safety and quality for raw materials are not prioritized. In this case, research-use only products can be used. However, when progressing into later drug development stages such as CMC or clinical phases, it is necessary to replace the raw materials with GMP-grade materials to be compliant with regulatory guidelines. In this transitional period, there is a significant amount of energy dedicated to re-evaluating and validating new raw materials.
To ease this transition from the preclinical development into the clinical stage, we offer several grades of cytokines that have been evaluated to be near-identical in bioactivity, along with the documentation required from GMP products. We recommend using our Premium(Pre-GMP) grade raw materials in the early development stage to seamlessly transition to our GMP-grade raw materials when entering CMC or clinical phases and minimize the number of re-evaluation and validation studies performed.
To support ongoing cell therapy manufacturers and developers, ACROBiosystems has developed a wide array of high-quality cytokines for the in vitro culture of immune cells, stem cells, organoids, and various other cell types. We also offer Premium(Pre-GMP) and GMP grades for our cytokines to better meet the needs of various drug development stages and applications. All the cytokines are manufactured using a similar production process, therefore enabling a seamless transition between grades. As a result, we enable you to seamlessly transition between our products and accelerate your research and development.
| Premium(Pre-GMP) Grade | GMP Grade | |
|---|---|---|
| Application | Research and Development; Preclinical research, seamless transition into clinical phases | Designed to meet clinical phase requirements and bolster your IND application to various regulatory bodies. |
| Quality System | ISO 9001 /ISO 13485 Certified | ISO 9001 /ISO 13485 Certified (Development stage) GMP Quality Management System (Production stage) |
| Production | ISO certified facilities | GMP certified facilities |
| Transient or stable cell lines | Stable cell lines (Full inspection according to USP and ChP) | |
| Animal-origin free materials or BSE/TSE free | Animal-origin free materials or BSE/TSE free | |
| Pharmaceutical-grade materials | Pharmaceutical-grade materials | |
| Strict sterilization filtration | Strict sterilization filtration | |
| Class C+A room with manual aseptic filling (ISO5) | B+A Cleanroom aseptic filling | |
| No additional virus clearance steps | Additional specific virus removal processes | |
| Quality Control | Sterility / Mycoplasma testing | Sterility / Mycoplasma testing |
| Endotoxin control and detection | Endotoxin control and detection | |
| Validated key equipment and analytical instruments | Validated equipment /analytical instruments/analytical methods(Audit trail available) | |
| Residual DNA/HCP testing | Residual DNA/HCP testing | |
| Limited adventitious agent testing | Stricter standards for viral residues alongside animal safety evaluations (select products). | |
| Documentation | Common regulatory support | Comprehensive regulatory support files |
| DMF filing (Few products) | DMF filing (All products) |

Transitioning from Premium(Pre-GMP) to GMP 
Cytokines are critical raw materials used in cell culturing for cell and gene therapy (CGT) drugs. Usually, in the preclinical stage, both safety and quality for raw materials are not prioritized. In this case, research-use only products can be used. However, when progressing into later drug development stages such as CMC or clinical phases, it is necessary to replace the raw materials with GMP-grade materials to be compliant with regulatory guidelines. In this transitional period, there is a significant amount of energy dedicated to re-evaluating and validating new raw materials.
To ease this transition from the preclinical development into the clinical stage, we offer several grades of cytokines that have been evaluated to be near-identical in bioactivity, along with the documentation required from GMP products. We recommend using our Premium(Pre-GMP) grade raw materials in the early development stage to seamlessly transition to our GMP-grade raw materials when entering CMC or clinical phases and minimize the number of re-evaluation and validation studies performed.
To support ongoing cell therapy manufacturers and developers, ACROBiosystems has developed a wide array of high-quality cytokines for the in vitro culture of immune cells, stem cells, organoids, and various other cell types. We also offer Premium(Pre-GMP) and GMP grades for our cytokines to better meet the needs of various drug development stages and applications. All the cytokines are manufactured using a similar production process, therefore enabling a seamless transition between grades. As a result, we enable you to seamlessly transition between our products and accelerate your research and development.
| Premium(Pre-GMP) Grade | GMP Grade | |
|---|---|---|
| Application | Research and Development; Preclinical research, seamless transition into clinical phases | Designed to meet clinical phase requirements and bolster your IND application to various regulatory bodies. |
| Quality System | ISO 9001 /ISO 13485 Certified | ISO 9001 /ISO 13485 Certified (Development stage) GMP Quality Management System (Production stage) |
| Production | ISO certified facilities | GMP certified facilities |
| Transient or stable cell lines | Stable cell lines (Full inspection according to USP and ChP) | |
| Animal-origin free materials or BSE/TSE free | Animal-origin free materials or BSE/TSE free | |
| Pharmaceutical-grade materials | Pharmaceutical-grade materials | |
| Strict sterilization filtration | Strict sterilization filtration | |
| Class C+A room with manual aseptic filling (ISO5) | B+A Cleanroom aseptic filling | |
| No additional virus clearance steps | Additional specific virus removal processes | |
| Quality Control | Sterility / Mycoplasma testing | Sterility / Mycoplasma testing |
| Endotoxin control and detection | Endotoxin control and detection | |
| Validated key equipment and analytical instruments | Validated equipment /analytical instruments/analytical methods(Audit trail available) | |
| Residual DNA/HCP testing | Residual DNA/HCP testing | |
| Limited adventitious agent testing | Stricter standards for viral residues alongside animal safety evaluations (select products). | |
| Documentation | Common regulatory support | Comprehensive regulatory support files |
| DMF filing (Few products) | DMF filing (All products) |

Transitioning from Premium(Pre-GMP) to GMP 
Cytokines are critical raw materials used in cell culturing for cell and gene therapy (CGT) drugs. Usually, in the preclinical stage, both safety and quality for raw materials are not prioritized. In this case, research-use only products can be used. However, when progressing into later drug development stages such as CMC or clinical phases, it is necessary to replace the raw materials with GMP-grade materials to be compliant with regulatory guidelines. In this transitional period, there is a significant amount of energy dedicated to re-evaluating and validating new raw materials.
To ease this transition from the preclinical development into the clinical stage, we offer several grades of cytokines that have been evaluated to be near-identical in bioactivity, along with the documentation required from GMP products. We recommend using our Premium(Pre-GMP) grade raw materials in the early development stage to seamlessly transition to our GMP-grade raw materials when entering CMC or clinical phases and minimize the number of re-evaluation and validation studies performed.