- English
- 日本語
- 한국어
- Deutsch
- Français
- Español
No data
官方服务号
招聘公众号
The essence of Resilient Supply is thoroughly embedded in the design and operational philosophy of ACROBiosystems' GMP facility. The facility features intelligent and modular design, boasting flexibility and resilience, capable of tackling complex and diverse production tasks to meet various supply demands for GMP-grade critical reagents.
Providing high-quality GMP products at an affordable cost can help lower the barrier in accessing innovative medicines including cell therapies. Our new facility was designed specifically for GMP production of proteins, enzymes, activation beads, and other solutions.
Want to learn more about
your GMP Protein options?
Quality control testing throughout the entire production process starting from bulk intermediates to the final lyophilized product ensures that our GMP grade products meets your needs and remains compliant. We ensure bioactivity, consistency, and stability in all our GMP solutions through our comprehensive quality management system.
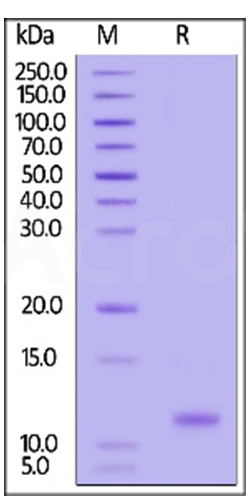
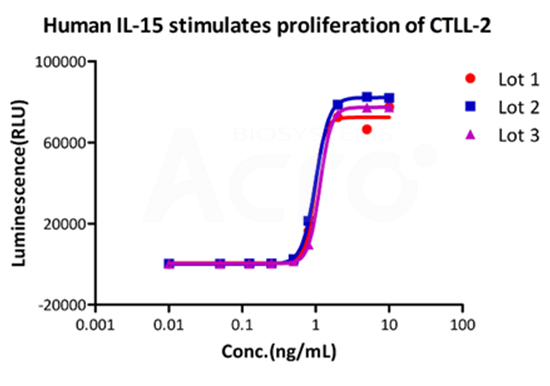
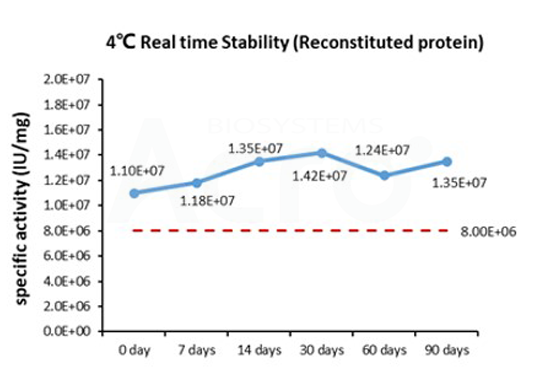
Three independent lots of GMP Human IL-15 (Cat. No. GMP-L15H13) were tested for the ability to simulate the proliferation of CTLL-2 cells. Average specific activity of GMP Human IL-15 was defined to be more than 0.8 x 107 IU/mg after calibration against human IL-15 WHO International Standard (NIBSC code: 95/554).
Human IL-2, GMP-grade
Human IL-7, GMP-grade
Human IL-15, GMP-grade
Explore >>
Request a protocol for GMP-grade IL-15 Bioactivity Verification
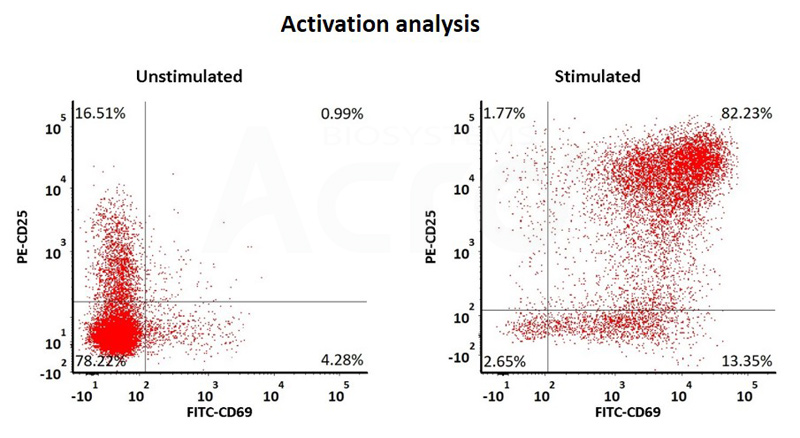
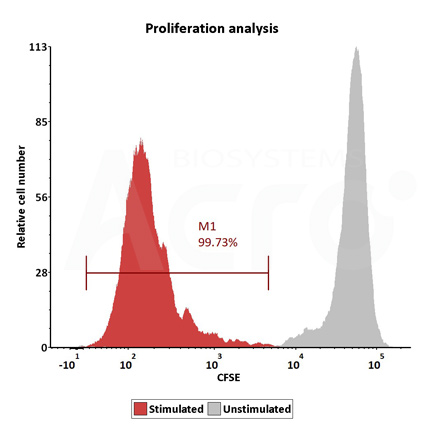
Human T cells were stimulated using GMP-grade ActiveMax Human T Cell Activation / Expansion CD3/CD28 beads (Cat. No. GMP-MBS001 ) for 24 hours. Activation was assessed by measuring expression of both activation markers CD25 and CD69 expression on T cell surface by staining with PE labeled anti-human CD25 antibody and FITC-labeled anti-human CD69 antibody respectively (QC tested).
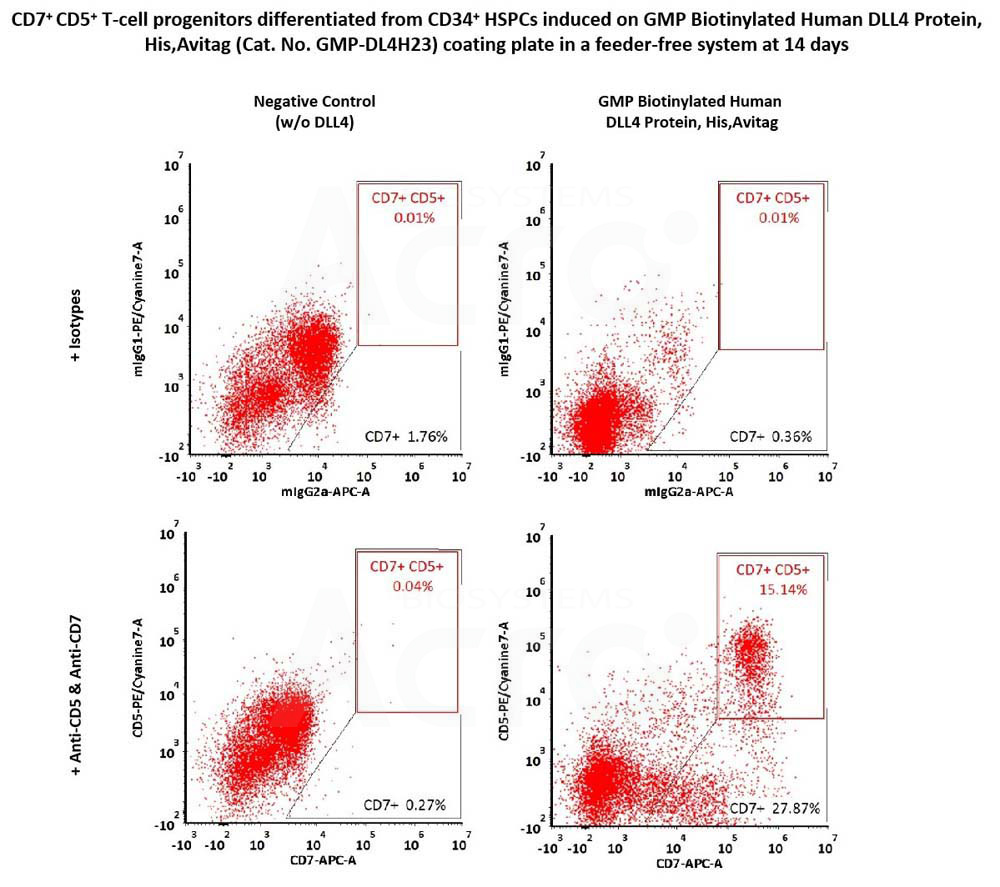
CD34+ CD45+ hematopoietic cells were seeded on GMP Biotinylated Human DLL4 Protein, His,Avitag (Cat. No. GMP-DL4H23) coated plates and differentiated for 14 days, then flow cytometry was used to detect the expression of T-cell progenitor markers, CD7 and CD5. The GMP Biotinylated Human DLL4 Protein, His,Avitag together with SCF, TPO, Flt3L and IL7, could induce the formation of CD7+ and CD7+ CD5+ T-cell progenitors (Routinely tested).
DLL4, Fc tag, GMP-grade
Biotinylated DLL4, His, Avitag, GMP-grade
VCAM-1, GMP-grade
Explore >>

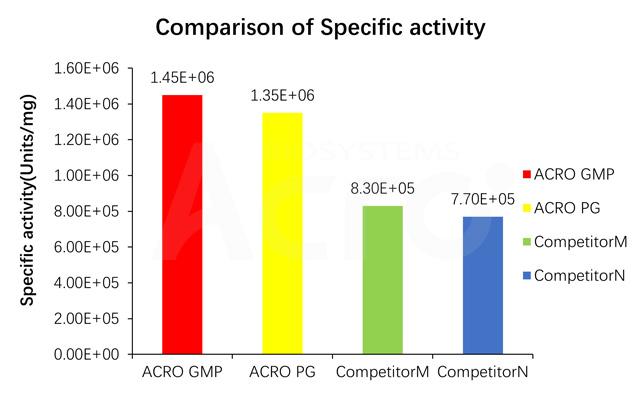
Specific activity for GMP GENIUS Nuclease is measured under standard assay conditions. The specific activity of GMP GENIUS Nuclease, is >1.2E+06 unit/mg protein. One unit will digest sonicated salmon sperm DNA to acid-soluble oligonucleotides equivalent to a ΔA260 of 1.0 in 30 min at pH 8.0 at 37 °C, which corresponds approximately to complete digestion of 37 μg DNA.
Salt-Active GENIUS Nuclease, GMP-grade
NLS-Cas9 Nuclease, GMP-grade
Explore >>
Interested in testing
a GMP grade product ?
The essence of Resilient Supply is thoroughly embedded in the design and operational philosophy of ACROBiosystems' GMP facility. The facility features intelligent and modular design, boasting flexibility and resilience, capable of tackling complex and diverse production tasks to meet various supply demands for GMP-grade critical reagents.
Providing high-quality GMP products at an affordable cost can help lower the barrier in accessing innovative medicines including cell therapies. Our new facility was designed specifically for GMP production of proteins, enzymes, activation beads, and other solutions.
Want to learn more about
your GMP Protein options?
Quality control testing throughout the entire production process starting from bulk intermediates to the final lyophilized product ensures that our GMP grade products meets your needs and remains compliant. We ensure bioactivity, consistency, and stability in all our GMP solutions through our comprehensive quality management system.



Three independent lots of GMP Human IL-15 (Cat. No. GMP-L15H13) were tested for the ability to simulate the proliferation of CTLL-2 cells. Average specific activity of GMP Human IL-15 was defined to be more than 0.8 x 107 IU/mg after calibration against human IL-15 WHO International Standard (NIBSC code: 95/554).
Human IL-2, GMP-grade
Human IL-7, GMP-grade
Human IL-15, GMP-grade
Explore >>
Request a protocol for GMP-grade IL-15 Bioactivity Verification


Human T cells were stimulated using GMP-grade ActiveMax Human T Cell Activation / Expansion CD3/CD28 beads (Cat. No. GMP-MBS001 ) for 24 hours. Activation was assessed by measuring expression of both activation markers CD25 and CD69 expression on T cell surface by staining with PE labeled anti-human CD25 antibody and FITC-labeled anti-human CD69 antibody respectively (QC tested).

CD34+ CD45+ hematopoietic cells were seeded on GMP Biotinylated Human DLL4 Protein, His,Avitag (Cat. No. GMP-DL4H23) coated plates and differentiated for 14 days, then flow cytometry was used to detect the expression of T-cell progenitor markers, CD7 and CD5. The GMP Biotinylated Human DLL4 Protein, His,Avitag together with SCF, TPO, Flt3L and IL7, could induce the formation of CD7+ and CD7+ CD5+ T-cell progenitors (Routinely tested).
DLL4, Fc tag, GMP-grade
Biotinylated DLL4, His, Avitag, GMP-grade
VCAM-1, GMP-grade
Explore >>


Specific activity for GMP GENIUS Nuclease is measured under standard assay conditions. The specific activity of GMP GENIUS Nuclease, is >1.2E+06 unit/mg protein. One unit will digest sonicated salmon sperm DNA to acid-soluble oligonucleotides equivalent to a ΔA260 of 1.0 in 30 min at pH 8.0 at 37 °C, which corresponds approximately to complete digestion of 37 μg DNA.
Salt-Active GENIUS Nuclease, GMP-grade
NLS-Cas9 Nuclease, GMP-grade
Explore >>
Interested in testing
a GMP grade product ?
The essence of Resilient Supply is thoroughly embedded in the design and operational philosophy of ACROBiosystems' GMP facility. The facility features intelligent and modular design, boasting flexibility and resilience, capable of tackling complex and diverse production tasks to meet various supply demands for GMP-grade critical reagents.
Providing high-quality GMP products at an affordable cost can help lower the barrier in accessing innovative medicines including cell therapies. Our new facility was designed specifically for GMP production of proteins, enzymes, activation beads, and other solutions.
Want to learn more about
your GMP Protein options?
Quality control testing throughout the entire production process starting from bulk intermediates to the final lyophilized product ensures that our GMP grade products meets your needs and remains compliant. We ensure bioactivity, consistency, and stability in all our GMP solutions through our comprehensive quality management system.



Three independent lots of GMP Human IL-15 (Cat. No. GMP-L15H13) were tested for the ability to simulate the proliferation of CTLL-2 cells. Average specific activity of GMP Human IL-15 was defined to be more than 0.8 x 107 IU/mg after calibration against human IL-15 WHO International Standard (NIBSC code: 95/554).
Human IL-2, GMP-grade
Human IL-7, GMP-grade
Human IL-15, GMP-grade
Explore >>
Request a protocol for GMP-grade IL-15 Bioactivity Verification


Human T cells were stimulated using GMP-grade ActiveMax Human T Cell Activation / Expansion CD3/CD28 beads (Cat. No. GMP-MBS001 ) for 24 hours. Activation was assessed by measuring expression of both activation markers CD25 and CD69 expression on T cell surface by staining with PE labeled anti-human CD25 antibody and FITC-labeled anti-human CD69 antibody respectively (QC tested).

CD34+ CD45+ hematopoietic cells were seeded on GMP Biotinylated Human DLL4 Protein, His,Avitag (Cat. No. GMP-DL4H23) coated plates and differentiated for 14 days, then flow cytometry was used to detect the expression of T-cell progenitor markers, CD7 and CD5. The GMP Biotinylated Human DLL4 Protein, His,Avitag together with SCF, TPO, Flt3L and IL7, could induce the formation of CD7+ and CD7+ CD5+ T-cell progenitors (Routinely tested).
DLL4, Fc tag, GMP-grade
Biotinylated DLL4, His, Avitag, GMP-grade
VCAM-1, GMP-grade
Explore >>


Specific activity for GMP GENIUS Nuclease is measured under standard assay conditions. The specific activity of GMP GENIUS Nuclease, is >1.2E+06 unit/mg protein. One unit will digest sonicated salmon sperm DNA to acid-soluble oligonucleotides equivalent to a ΔA260 of 1.0 in 30 min at pH 8.0 at 37 °C, which corresponds approximately to complete digestion of 37 μg DNA.
Salt-Active GENIUS Nuclease, GMP-grade
NLS-Cas9 Nuclease, GMP-grade
Explore >>
Interested in testing
a GMP grade product ?
The essence of Resilient Supply is thoroughly embedded in the design and operational philosophy of ACROBiosystems' GMP facility. The facility features intelligent and modular design, boasting flexibility and resilience, capable of tackling complex and diverse production tasks to meet various supply demands for GMP-grade critical reagents.
Providing high-quality GMP products at an affordable cost can help lower the barrier in accessing innovative medicines including cell therapies. Our new facility was designed specifically for GMP production of proteins, enzymes, activation beads, and other solutions.
Want to learn more about
your GMP Protein options?
Quality control testing throughout the entire production process starting from bulk intermediates to the final lyophilized product ensures that our GMP grade products meets your needs and remains compliant. We ensure bioactivity, consistency, and stability in all our GMP solutions through our comprehensive quality management system.



Three independent lots of GMP Human IL-15 (Cat. No. GMP-L15H13) were tested for the ability to simulate the proliferation of CTLL-2 cells. Average specific activity of GMP Human IL-15 was defined to be more than 0.8 x 107 IU/mg after calibration against human IL-15 WHO International Standard (NIBSC code: 95/554).
Human IL-2, GMP-grade
Human IL-7, GMP-grade
Human IL-15, GMP-grade
Explore >>
Request a protocol for GMP-grade IL-15 Bioactivity Verification


Human T cells were stimulated using GMP-grade ActiveMax Human T Cell Activation / Expansion CD3/CD28 beads (Cat. No. GMP-MBS001 ) for 24 hours. Activation was assessed by measuring expression of both activation markers CD25 and CD69 expression on T cell surface by staining with PE labeled anti-human CD25 antibody and FITC-labeled anti-human CD69 antibody respectively (QC tested).

CD34+ CD45+ hematopoietic cells were seeded on GMP Biotinylated Human DLL4 Protein, His,Avitag (Cat. No. GMP-DL4H23) coated plates and differentiated for 14 days, then flow cytometry was used to detect the expression of T-cell progenitor markers, CD7 and CD5. The GMP Biotinylated Human DLL4 Protein, His,Avitag together with SCF, TPO, Flt3L and IL7, could induce the formation of CD7+ and CD7+ CD5+ T-cell progenitors (Routinely tested).
DLL4, Fc tag, GMP-grade
Biotinylated DLL4, His, Avitag, GMP-grade
VCAM-1, GMP-grade
Explore >>


Specific activity for GMP GENIUS Nuclease is measured under standard assay conditions. The specific activity of GMP GENIUS Nuclease, is >1.2E+06 unit/mg protein. One unit will digest sonicated salmon sperm DNA to acid-soluble oligonucleotides equivalent to a ΔA260 of 1.0 in 30 min at pH 8.0 at 37 °C, which corresponds approximately to complete digestion of 37 μg DNA.
Salt-Active GENIUS Nuclease, GMP-grade
NLS-Cas9 Nuclease, GMP-grade
Explore >>
Interested in testing
a GMP grade product ?
The essence of Resilient Supply is thoroughly embedded in the design and operational philosophy of ACROBiosystems' GMP facility. The facility features intelligent and modular design, boasting flexibility and resilience, capable of tackling complex and diverse production tasks to meet various supply demands for GMP-grade critical reagents.
Providing high-quality GMP products at an affordable cost can help lower the barrier in accessing innovative medicines including cell therapies. Our new facility was designed specifically for GMP production of proteins, enzymes, activation beads, and other solutions.
Want to learn more about
your GMP Protein options?
Quality control testing throughout the entire production process starting from bulk intermediates to the final lyophilized product ensures that our GMP grade products meets your needs and remains compliant. We ensure bioactivity, consistency, and stability in all our GMP solutions through our comprehensive quality management system.



Three independent lots of GMP Human IL-15 (Cat. No. GMP-L15H13) were tested for the ability to simulate the proliferation of CTLL-2 cells. Average specific activity of GMP Human IL-15 was defined to be more than 0.8 x 107 IU/mg after calibration against human IL-15 WHO International Standard (NIBSC code: 95/554).
Human IL-2, GMP-grade
Human IL-7, GMP-grade
Human IL-15, GMP-grade
Explore >>
Request a protocol for GMP-grade IL-15 Bioactivity Verification


Human T cells were stimulated using GMP-grade ActiveMax Human T Cell Activation / Expansion CD3/CD28 beads (Cat. No. GMP-MBS001 ) for 24 hours. Activation was assessed by measuring expression of both activation markers CD25 and CD69 expression on T cell surface by staining with PE labeled anti-human CD25 antibody and FITC-labeled anti-human CD69 antibody respectively (QC tested).

CD34+ CD45+ hematopoietic cells were seeded on GMP Biotinylated Human DLL4 Protein, His,Avitag (Cat. No. GMP-DL4H23) coated plates and differentiated for 14 days, then flow cytometry was used to detect the expression of T-cell progenitor markers, CD7 and CD5. The GMP Biotinylated Human DLL4 Protein, His,Avitag together with SCF, TPO, Flt3L and IL7, could induce the formation of CD7+ and CD7+ CD5+ T-cell progenitors (Routinely tested).
DLL4, Fc tag, GMP-grade
Biotinylated DLL4, His, Avitag, GMP-grade
VCAM-1, GMP-grade
Explore >>


Specific activity for GMP GENIUS Nuclease is measured under standard assay conditions. The specific activity of GMP GENIUS Nuclease, is >1.2E+06 unit/mg protein. One unit will digest sonicated salmon sperm DNA to acid-soluble oligonucleotides equivalent to a ΔA260 of 1.0 in 30 min at pH 8.0 at 37 °C, which corresponds approximately to complete digestion of 37 μg DNA.
Salt-Active GENIUS Nuclease, GMP-grade
NLS-Cas9 Nuclease, GMP-grade
Explore >>
Interested in testing
a GMP grade product ?
Search for the solutions that fit your needs by your application! We currently offer a wide array of products designed to help with cell manufacturing of immune cells including T cells, NK cells, and Dendritic cells. We also offer an increasingly large catalog of iPSC growth factors and differentiation factors. This includes recombinant proteins such as DLL4, VCAM-1, and many others. Basement membrane extracts and extracellular matrix proteins are also available.
Know exactly what you’re looking for? Explore our catalog of solutions to find what’s right for you. Our GMP-grade solutions are manufactured under regulatory guidelines for ancillary materials in cell therapy manufacturing processes. Related Premium (Pre-GMP) grade solutions are also listed to help you find related products to help you streamline your transition to GMP.
Learn More About our Transitioning to GMP grade Solutions 
Learn More About our GMP Capabilities, Quality, and Regulatory Support 
| Molecule | Source | GMP Grade Catalog No. | DMF Filed for GMP Grade | Premium (Pre-GMP) Grade Catalog No. |
|---|
*Already purchased a DMF-filed protein? Click here to apply for your DMF authorization.
Search for the solutions that fit your needs by your application! We currently offer a wide array of products designed to help with cell manufacturing of immune cells including T cells, NK cells, and Dendritic cells. We also offer an increasingly large catalog of iPSC growth factors and differentiation factors. This includes recombinant proteins such as DLL4, VCAM-1, and many others. Basement membrane extracts and extracellular matrix proteins are also available.
Know exactly what you’re looking for? Explore our catalog of solutions to find what’s right for you. Our GMP-grade solutions are manufactured under regulatory guidelines for ancillary materials in cell therapy manufacturing processes. Related Premium (Pre-GMP) grade solutions are also listed to help you find related products to help you streamline your transition to GMP.
Learn More About our Transitioning to GMP grade Solutions 
Learn More About our GMP Capabilities, Quality, and Regulatory Support 
| Molecule | Source | GMP Grade Catalog No. | DMF Filed for GMP Grade | Premium (Pre-GMP) Grade Catalog No. |
|---|
*Already purchased a DMF-filed protein? Click here to apply for your DMF authorization.
Search for the solutions that fit your needs by your application! We currently offer a wide array of products designed to help with cell manufacturing of immune cells including T cells, NK cells, and Dendritic cells. We also offer an increasingly large catalog of iPSC growth factors and differentiation factors. This includes recombinant proteins such as DLL4, VCAM-1, and many others. Basement membrane extracts and extracellular matrix proteins are also available.
Know exactly what you’re looking for? Explore our catalog of solutions to find what’s right for you. Our GMP-grade solutions are manufactured under regulatory guidelines for ancillary materials in cell therapy manufacturing processes. Related Premium (Pre-GMP) grade solutions are also listed to help you find related products to help you streamline your transition to GMP.
Learn More About our Transitioning to GMP grade Solutions 
Learn More About our GMP Capabilities, Quality, and Regulatory Support 
| Molecule | Source | GMP Grade Catalog No. | DMF Filed for GMP Grade | Premium (Pre-GMP) Grade Catalog No. |
|---|
*Already purchased a DMF-filed protein? Click here to apply for your DMF authorization.
Search for the solutions that fit your needs by your application! We currently offer a wide array of products designed to help with cell manufacturing of immune cells including T cells, NK cells, and Dendritic cells. We also offer an increasingly large catalog of iPSC growth factors and differentiation factors. This includes recombinant proteins such as DLL4, VCAM-1, and many others. Basement membrane extracts and extracellular matrix proteins are also available.
Know exactly what you’re looking for? Explore our catalog of solutions to find what’s right for you. Our GMP-grade solutions are manufactured under regulatory guidelines for ancillary materials in cell therapy manufacturing processes. Related Premium (Pre-GMP) grade solutions are also listed to help you find related products to help you streamline your transition to GMP.
Learn More About our Transitioning to GMP grade Solutions 
Learn More About our GMP Capabilities, Quality, and Regulatory Support 
| Molecule | Source | GMP Grade Catalog No. | DMF Filed for GMP Grade | Premium (Pre-GMP) Grade Catalog No. |
|---|
*Already purchased a DMF-filed protein? Click here to apply for your DMF authorization.