产品详情
分子别名(Synonym)
Glycoprotein E/gE (VZV)
表达区间及表达系统(Source)
Varicella zoster virus (strain Oka vaccine) Envelope Glycoprotein E (gE), His Tag (GLE-V52H3) is expressed from human 293 cells (HEK293). It contains AA Ser 31 - Ala 546 (Accession # Q9J3M8-1).
Predicted N-terminus: Ser 31
Request for sequence蛋白结构(Molecular Characterization)

This protein carries a polyhistidine tag at the C-terminus.
The protein has a calculated MW of 60.2 kDa. The protein migrates as 54-69 kDa when calibrated against Star Ribbon Pre-stained Protein Marker under non-reducing (NR) condition (SDS-PAGE) due to glycosylation.
偶联(Conjugate)
Unconjugated
内毒素(Endotoxin)
Less than 1.0 EU per μg by the LAL method.
纯度(Purity)
90% as determined by SDS-PAGE.
90% as determined by SEC-MALS.
制剂(Formulation)
Lyophilized from 0.22 μm filtered solution in PBS, pH7.4 with trehalose as protectant.
Contact us for customized product form or formulation.
重构方法(Reconstitution)
Please see Certificate of Analysis for specific instructions.
For best performance, we strongly recommend you to follow the reconstitution protocol provided in the CoA.
存储(Storage)
For long term storage, the product should be stored at lyophilized state at -20°C or lower.
Please avoid repeated freeze-thaw cycles.
This product is stable after storage at:
- -20°C to -70°C for 12 months in lyophilized state;
- -70°C for 3 months under sterile conditions after reconstitution.
质量管理控制体系(QMS)
产品展示
电泳(SDS-PAGE)
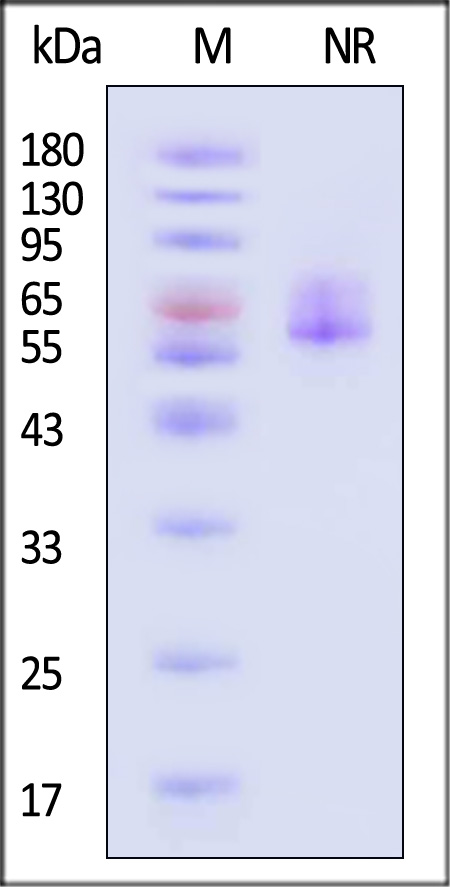
Varicella zoster virus (strain Oka vaccine) Envelope Glycoprotein E (gE), His Tag on SDS-PAGE under non-reducing (NR) condition. The gel was stained with Coomassie Blue. The purity of the protein is greater than 90% (With Star Ribbon Pre-stained Protein Marker).
SEC-MALS
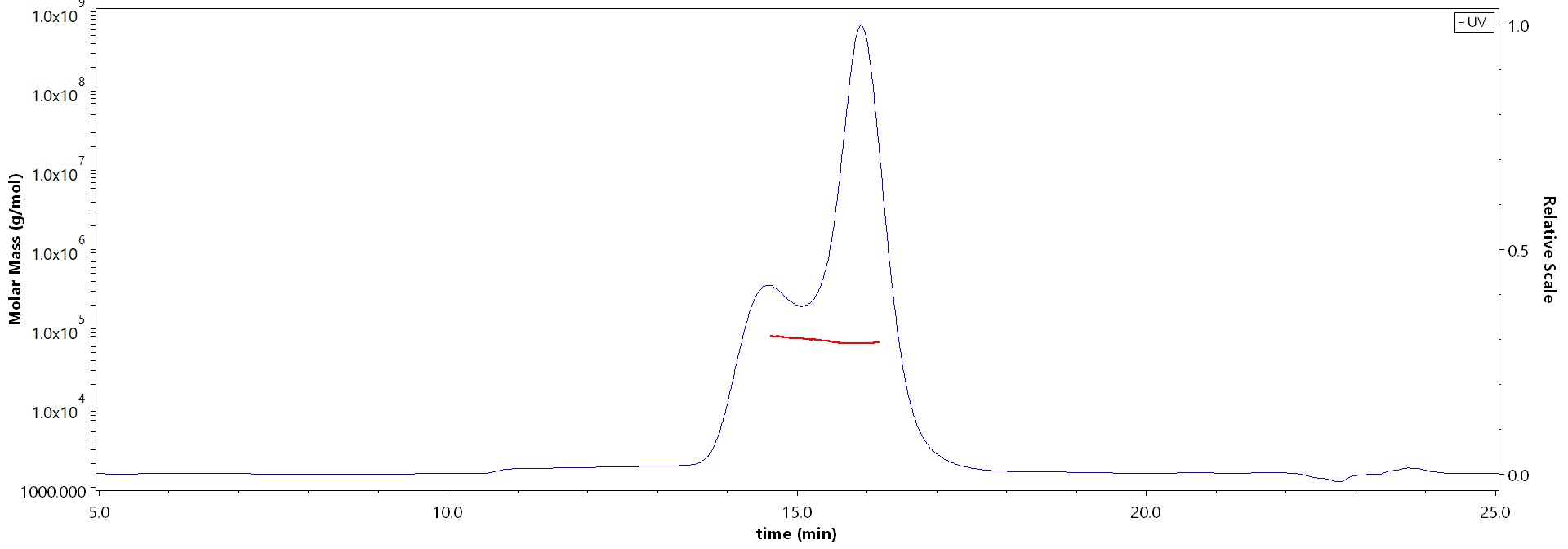
The purity of Varicella zoster virus (strain Oka vaccine) Envelope Glycoprotein E (gE), His Tag (Cat. No. GLE-V52H3) is more than 90% and the molecular weight of this protein is around 60-85 kDa verified by SEC-MALS.
Report
活性(Bioactivity)-ELISA
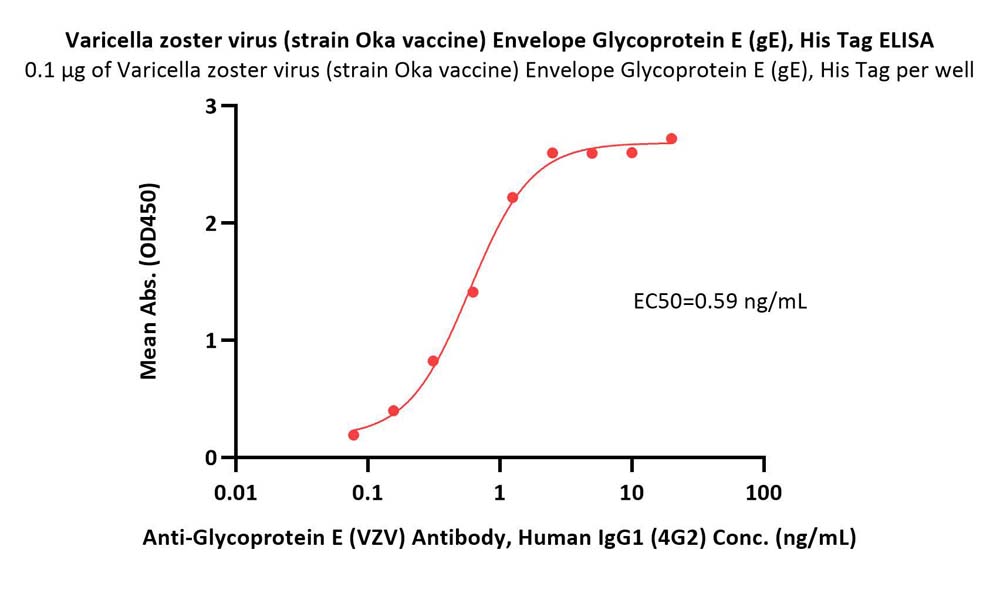
Immobilized Varicella zoster virus (strain Oka vaccine) Envelope Glycoprotein E (gE), His Tag (Cat. No. GLE-V52H3) at 1 μg/mL (100 μL/well) can bind Anti-Glycoprotein E (VZV) Antibody, Human IgG1 (4G2) with a linear range of 0.08-1 ng/mL (QC tested).
Protocol
活性(Bioactivity)-SPR
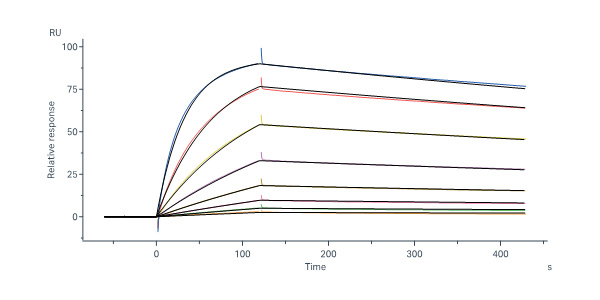
Anti-Glycoprotein E (VZV) Antibody, Human IgG1 (4G2) captured on Protein A Chip can bind Varicella zoster virus (strain Oka vaccine) Envelope Glycoprotein E (gE), His Tag (Cat. No. GLE-V52H3) with an affinity constant of 4.81 nM as determined in SPR assay (Biacore 8K) (Routinely tested).
Protocol
用户评价 发表评论

背景介绍
Glycoprotein E (gE) is one of the known glycoproteins (gB, gC, gE, gH, gI, gK, gI) of VZV that is most abundantly expressed on the surface of virus and infected cells, playing an important role in viral replication and cell-to-cell spread. The strongly immunogenic gE can provide strong IgG signal in body fluid, which makes it ideal to be developed as an antigen for analysis of IgG antibodies. gE also demonstrates high potency as a vaccine immunogen and is formulated as the single viral envelope protein that constitutes the GSK VZV recombinant subunit vaccine Shingrix®.