- English
- 日本語
- 한국어
- Deutsch
- Français
- Español
No data
官方服务号
招聘公众号

| Principle of SEC-MALS |
|---|
| Size-exclusion Chromatography (SEC) is a chromatographic method that separates components by its molecular size. Uniformly porous beads are packed into a chromatographic column to separate a sample based on molecular size, and the protein purity is obtained according to the separation spectrum. Each component separated by SEC enters the multi-angle light scattering (MALS), and light scattering occurs when the laser illuminates each group. MALS is a post-column detection method that measures the intensity of scattered light at multiple angles at the same time, and the intensity is proportional to the square of molar mass, concentration and refractive index increment. Through the combination of SEC-MALS, the molecular weight of an analyte can be directly calculated and the aggregation form of protein can be judged. |
| Sample Type |
|---|
| ※ Recombinant Proteins ※ Antibodies ※ Polypeptides (Column and method shall be provided) |

| Principle of UNcle |
|---|
| UNcle is a technology platform that tracks the folding state of protein by detecting the changes in its endogenous fluorescence through full-wavelength fluorescence. This determines the Tm of the protein stability and reflects the thermal stability or chemical stability of the protein. Using Static light scattering (SLS) , the stability of protein aggregation can be calculated. During the process of protein denaturation, its conformation gradually opens and hydrophobic points are gradually exposed, and when a certain level (temperature) is reached, the protein molecules begin to aggregate, leading to an increase in scattered light signal, which determines the initial Tagg of the protein. Using dynamic light scattering (DLS), the particle size and its distribution can be calculated through the fluctuation and the correlation function of light intensity. |
| Sample Type |
|---|
| ※ Tm&Tagg determination: Recombinant Proteins, Monoclonal Antibodies, Bispecific Antibodies, Antibody-Drug Conjugate (ADC), Adeno-Associated Virus (AAV), Lipid Nanoparticle (LNP) ※ Particle size determination:Unlimited |

| Principle of SEC-MALS |
|---|
| Size-exclusion Chromatography (SEC) is a chromatographic method that separates components by its molecular size. Uniformly porous beads are packed into a chromatographic column to separate a sample based on molecular size, and the protein purity is obtained according to the separation spectrum. Each component separated by SEC enters the multi-angle light scattering (MALS), and light scattering occurs when the laser illuminates each group. MALS is a post-column detection method that measures the intensity of scattered light at multiple angles at the same time, and the intensity is proportional to the square of molar mass, concentration and refractive index increment. Through the combination of SEC-MALS, the molecular weight of an analyte can be directly calculated and the aggregation form of protein can be judged. |
| Sample Type |
|---|
| ※ Recombinant Proteins ※ Antibodies ※ Polypeptides (Column and method shall be provided) |

| Principle of UNcle |
|---|
| UNcle is a technology platform that tracks the folding state of protein by detecting the changes in its endogenous fluorescence through full-wavelength fluorescence. This determines the Tm of the protein stability and reflects the thermal stability or chemical stability of the protein. Using Static light scattering (SLS) , the stability of protein aggregation can be calculated. During the process of protein denaturation, its conformation gradually opens and hydrophobic points are gradually exposed, and when a certain level (temperature) is reached, the protein molecules begin to aggregate, leading to an increase in scattered light signal, which determines the initial Tagg of the protein. Using dynamic light scattering (DLS), the particle size and its distribution can be calculated through the fluctuation and the correlation function of light intensity. |
| Sample Type |
|---|
| ※ Tm&Tagg determination: Recombinant Proteins, Monoclonal Antibodies, Bispecific Antibodies, Antibody-Drug Conjugate (ADC), Adeno-Associated Virus (AAV), Lipid Nanoparticle (LNP) ※ Particle size determination:Unlimited |

| Principle of SEC-MALS |
|---|
| Size-exclusion Chromatography (SEC) is a chromatographic method that separates components by its molecular size. Uniformly porous beads are packed into a chromatographic column to separate a sample based on molecular size, and the protein purity is obtained according to the separation spectrum. Each component separated by SEC enters the multi-angle light scattering (MALS), and light scattering occurs when the laser illuminates each group. MALS is a post-column detection method that measures the intensity of scattered light at multiple angles at the same time, and the intensity is proportional to the square of molar mass, concentration and refractive index increment. Through the combination of SEC-MALS, the molecular weight of an analyte can be directly calculated and the aggregation form of protein can be judged. |
| Sample Type |
|---|
| ※ Recombinant Proteins ※ Antibodies ※ Polypeptides (Column and method shall be provided) |

| Principle of UNcle |
|---|
| UNcle is a technology platform that tracks the folding state of protein by detecting the changes in its endogenous fluorescence through full-wavelength fluorescence. This determines the Tm of the protein stability and reflects the thermal stability or chemical stability of the protein. Using Static light scattering (SLS) , the stability of protein aggregation can be calculated. During the process of protein denaturation, its conformation gradually opens and hydrophobic points are gradually exposed, and when a certain level (temperature) is reached, the protein molecules begin to aggregate, leading to an increase in scattered light signal, which determines the initial Tagg of the protein. Using dynamic light scattering (DLS), the particle size and its distribution can be calculated through the fluctuation and the correlation function of light intensity. |
| Sample Type |
|---|
| ※ Tm&Tagg determination: Recombinant Proteins, Monoclonal Antibodies, Bispecific Antibodies, Antibody-Drug Conjugate (ADC), Adeno-Associated Virus (AAV), Lipid Nanoparticle (LNP) ※ Particle size determination:Unlimited |
 Determination of absolute molecular weight
Determination of absolute molecular weight  Confirmation of degree of polymerization
Confirmation of degree of polymerization  Determination Tm&Tagg
Determination Tm&Tagg  Determination of particle size and distribution
Determination of particle size and distribution SEC-MALS can determine the purity and degree of polymerization of various antibodies and proteins, while analyzing the modification of protein complexes by measuring the absolute molecular weight.
UNcle can carry out preparation development/optimization/formula screening, thermal stability evaluation and screening of drug candidate molecules (recombinant protein, monoclonal antibody, BsAb, ADC, AAV) by determining Tm&Tagg and particle size.
 Determination of absolute molecular weight
Determination of absolute molecular weight  Confirmation of degree of polymerization
Confirmation of degree of polymerization  Determination Tm&Tagg
Determination Tm&Tagg  Determination of particle size and distribution
Determination of particle size and distribution SEC-MALS can determine the purity and degree of polymerization of various antibodies and proteins, while analyzing the modification of protein complexes by measuring the absolute molecular weight.
UNcle can carry out preparation development/optimization/formula screening, thermal stability evaluation and screening of drug candidate molecules (recombinant protein, monoclonal antibody, BsAb, ADC, AAV) by determining Tm&Tagg and particle size.
 Determination of absolute molecular weight
Determination of absolute molecular weight  Confirmation of degree of polymerization
Confirmation of degree of polymerization  Determination Tm&Tagg
Determination Tm&Tagg  Determination of particle size and distribution
Determination of particle size and distribution SEC-MALS can determine the purity and degree of polymerization of various antibodies and proteins, while analyzing the modification of protein complexes by measuring the absolute molecular weight.
UNcle can carry out preparation development/optimization/formula screening, thermal stability evaluation and screening of drug candidate molecules (recombinant protein, monoclonal antibody, BsAb, ADC, AAV) by determining Tm&Tagg and particle size.
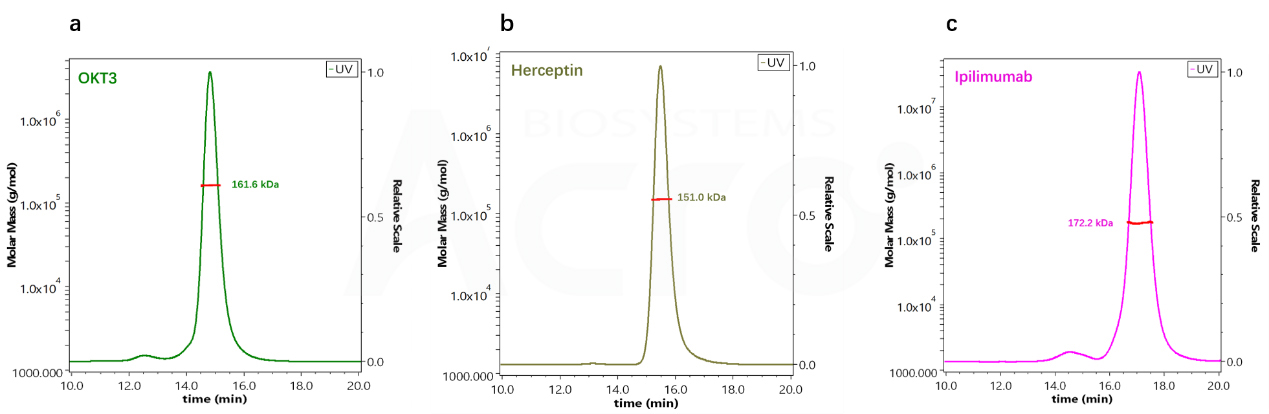
Three known antibody drugs, OKT3, Herceptin, and Ipilimumab, were determined by SEC-MALS. The molecular weight of the three antibodies is within a close range of 150-160 kDa. SEC-MALS was performed on the peaks with the retention time,calculating a molecular weight of 161.6, 15, and 172.2 kDa for each peak. The calculated molecular weight by SEC-MALS were close to the expected theoretical molecular weight (Fig. 1).
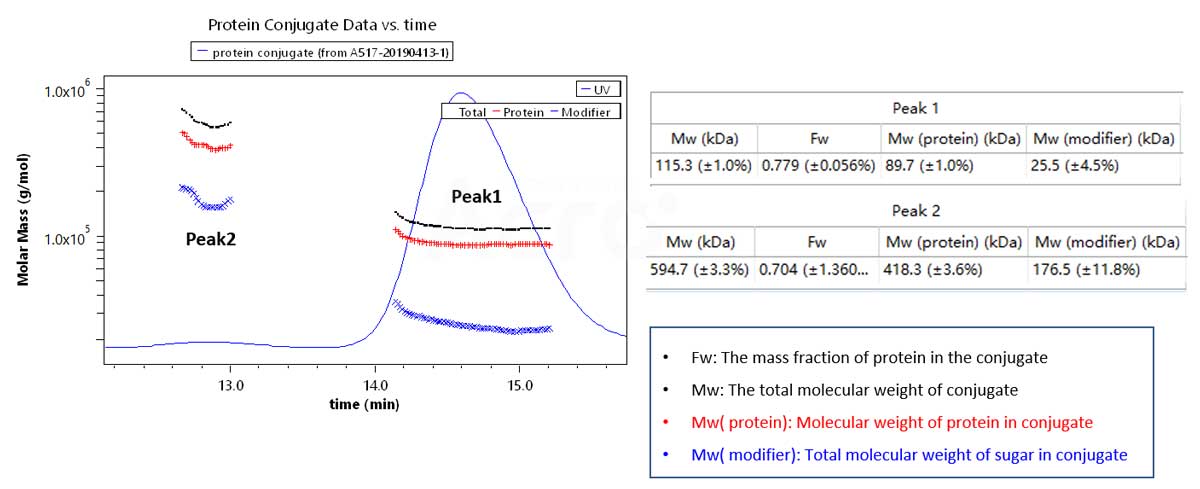
SEC-MALS can be used to determine glycosylation, PEG-modified proteins, membrane proteins and other complex modifications.
The degree of polymerization and protein purity is a critical part of protein characterization. For example, the SARS-CoV-2 virus S-protein functions as a trimer, consisting of three identical molecules encoded by the same gene. The researchers found that the conformational form of the S protein is naturally unstable, even if it does not bind to the host cell receptor ACE2. Similarly, there are also spontaneous conformational changes from prefusion to postfusion, which poses a signficiant challenges for vaccine development and neutralizing antibody screening. Cat. No. SPN-C52H9 (SARS-CoV-2 S protein, His Tag, Super stable trimer (MALS & NS-EM verified).
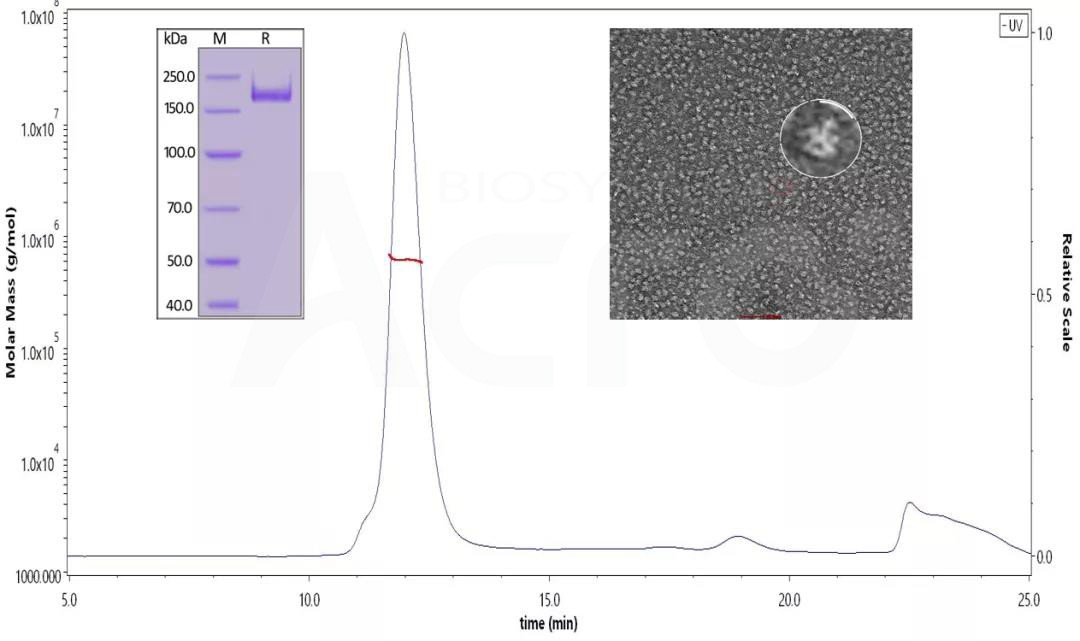
Fig 3. SDS-PAGE identified the SARS-CoV-2 S protein under reduced conditions. His Tag, Super stable trimer (MALS & NS-EM verified) (Cat. No. SPN-C52H9 ) The purity is over 90%, and the molecular weight is about 550-660 kDa by SEC-MALS. The size and appearance of the SARS-CoV-2 trimer verified with NS-EM were like those reported in the published literature.
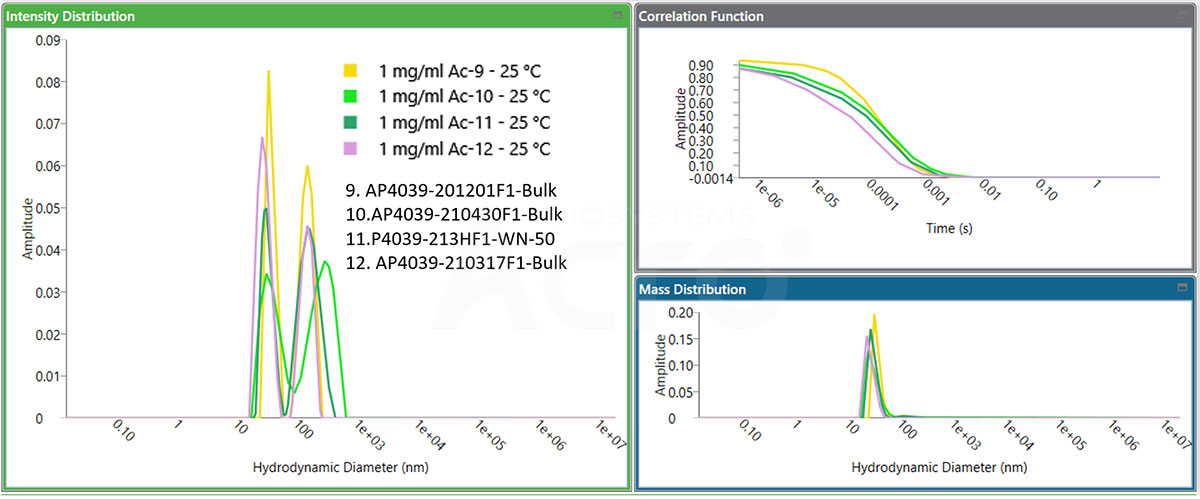
Candidate drug molecules can be screened based on their different initial conformations, which alters the calculated Tm values. As such, four different samples can be screened based on the calculated Tm.
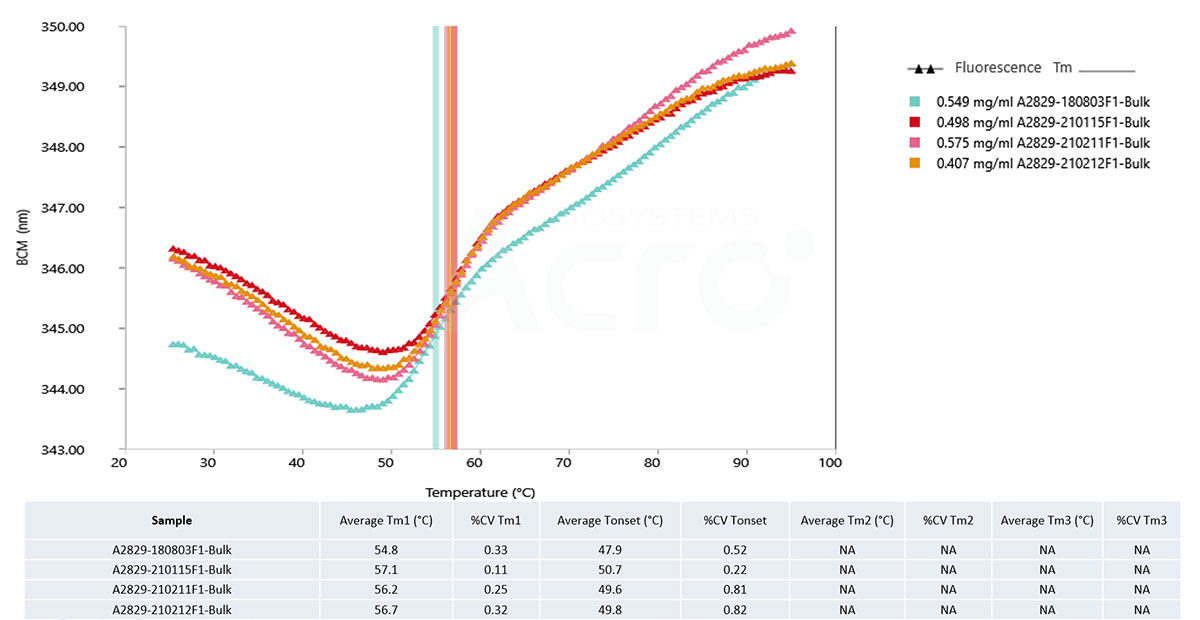
Fig 4. A2829-180803F1 Bulk Tm is relatively low, and the initial BCM value is significantly lower than that of the other three samples. It is speculated that its initial conformation state is different from that of the other three samples. The Tm detection value is also different from the other three samples.
Three known antibody drugs, OKT3, Herceptin, and Ipilimumab, were determined by SEC-MALS. The molecular weight of the three antibodies is within a close range of 150-160 kDa. SEC-MALS was performed on the peaks with the retention time,calculating a molecular weight of 161.6, 15, and 172.2 kDa for each peak. The calculated molecular weight by SEC-MALS were close to the expected theoretical molecular weight (Fig. 1).

SEC-MALS can be used to determine glycosylation, PEG-modified proteins, membrane proteins and other complex modifications.

The degree of polymerization and protein purity is a critical part of protein characterization. For example, the SARS-CoV-2 virus S-protein functions as a trimer, consisting of three identical molecules encoded by the same gene. The researchers found that the conformational form of the S protein is naturally unstable, even if it does not bind to the host cell receptor ACE2. Similarly, there are also spontaneous conformational changes from prefusion to postfusion, which poses a signficiant challenges for vaccine development and neutralizing antibody screening. Cat. No. SPN-C52H9 (SARS-CoV-2 S protein, His Tag, Super stable trimer (MALS & NS-EM verified).

Fig 3. SDS-PAGE identified the SARS-CoV-2 S protein under reduced conditions. His Tag, Super stable trimer (MALS & NS-EM verified) (Cat. No. SPN-C52H9 ) The purity is over 90%, and the molecular weight is about 550-660 kDa by SEC-MALS. The size and appearance of the SARS-CoV-2 trimer verified with NS-EM were like those reported in the published literature.
Candidate drug molecules can be screened based on their different initial conformations, which alters the calculated Tm values. As such, four different samples can be screened based on the calculated Tm.

Fig 4. A2829-180803F1 Bulk Tm is relatively low, and the initial BCM value is significantly lower than that of the other three samples. It is speculated that its initial conformation state is different from that of the other three samples. The Tm detection value is also different from the other three samples.

Three known antibody drugs, OKT3, Herceptin, and Ipilimumab, were determined by SEC-MALS. The molecular weight of the three antibodies is within a close range of 150-160 kDa. SEC-MALS was performed on the peaks with the retention time,calculating a molecular weight of 161.6, 15, and 172.2 kDa for each peak. The calculated molecular weight by SEC-MALS were close to the expected theoretical molecular weight (Fig. 1).

SEC-MALS can be used to determine glycosylation, PEG-modified proteins, membrane proteins and other complex modifications.

The degree of polymerization and protein purity is a critical part of protein characterization. For example, the SARS-CoV-2 virus S-protein functions as a trimer, consisting of three identical molecules encoded by the same gene. The researchers found that the conformational form of the S protein is naturally unstable, even if it does not bind to the host cell receptor ACE2. Similarly, there are also spontaneous conformational changes from prefusion to postfusion, which poses a signficiant challenges for vaccine development and neutralizing antibody screening. Cat. No. SPN-C52H9 (SARS-CoV-2 S protein, His Tag, Super stable trimer (MALS & NS-EM verified).

Fig 3. SDS-PAGE identified the SARS-CoV-2 S protein under reduced conditions. His Tag, Super stable trimer (MALS & NS-EM verified) (Cat. No. SPN-C52H9 ) The purity is over 90%, and the molecular weight is about 550-660 kDa by SEC-MALS. The size and appearance of the SARS-CoV-2 trimer verified with NS-EM were like those reported in the published literature.
Candidate drug molecules can be screened based on their different initial conformations, which alters the calculated Tm values. As such, four different samples can be screened based on the calculated Tm.

Fig 4. A2829-180803F1 Bulk Tm is relatively low, and the initial BCM value is significantly lower than that of the other three samples. It is speculated that its initial conformation state is different from that of the other three samples. The Tm detection value is also different from the other three samples.
