产品详情
优势特色(Features)
- Designed under ISO 9001:2015 and ISO 13485:2016
- Manufactured and QC tested under a GMP compliance factory
- FDA DMF filed
- Animal-Free materials
- Beta-lactam materials free
- Batch-to-batch consistency
- Stringent quality control tests
- No animal derived peptone and lactose used in production process
产品描述(Product Details)
GMP GENPower™ NLS-Cas9 Nuclease is a recombinant Streptococcus pyogenes Cas9 protein purified from Escherichia coli for CRISPR-based genome editing. The introduction of nuclear localization signals (NLS) can help Cas9 enter the nucleus, increasing the chance of genomic DNA cleavage.
应用说明(Application)
• Genetic modification of cells and gene therapy drugs (T cell, hematopoietic stem cell)
• High specificity detection of pathogens
浓度(Concentration)
10 mg/mL
纯度(Purity)
95% as determined by SDS-PAGE.
95% as determined by SEC-HPLC.
宿主蛋白残留(Host Cell Protein)
≤10 ng/mg of protein tested by ELISA.
宿主核酸残留(Host Cell DNA)
≤1 ng/mg of protein tested by qPCR.
无菌(Sterility)
The sterility testing was performed by membrane filtration method described in CP<1101>, USP<71> and Eur. Ph. 2.6.1.
内毒素(Endotoxin)
Less than 10 EU/mg by the LAL method.
制剂(Formulation)
Supplied as 0.2 μm filtered solution in 20 mM Tris, 300 mM NaCl, 0.1 mM EDTA, 1 mM TCEP, pH7.5.
Contact us for customized product form or formulation.
运输(Shipping)
This product is supplied and shipped with dry ice, please inquire the shipping cost.
存储(Storage)
This product is stable after storage at:
- The product MUST be stored at -20°C or lower upon receipt;
- -20°C for 5 years under sterile conditions.
产品展示
电泳(SDS-PAGE)
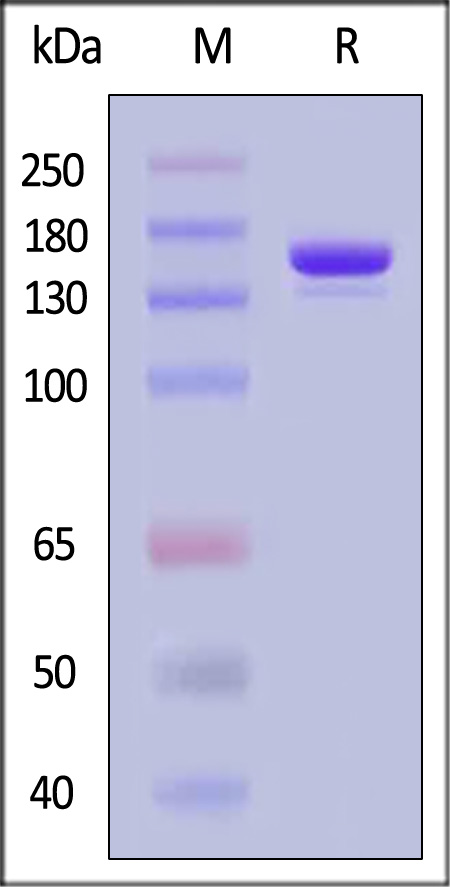
The gel was stained with Coomassie Blue. The purity of the protein is greater than 95% (With Star Ribbon Pre-stained Protein Marker).
SEC-MALS
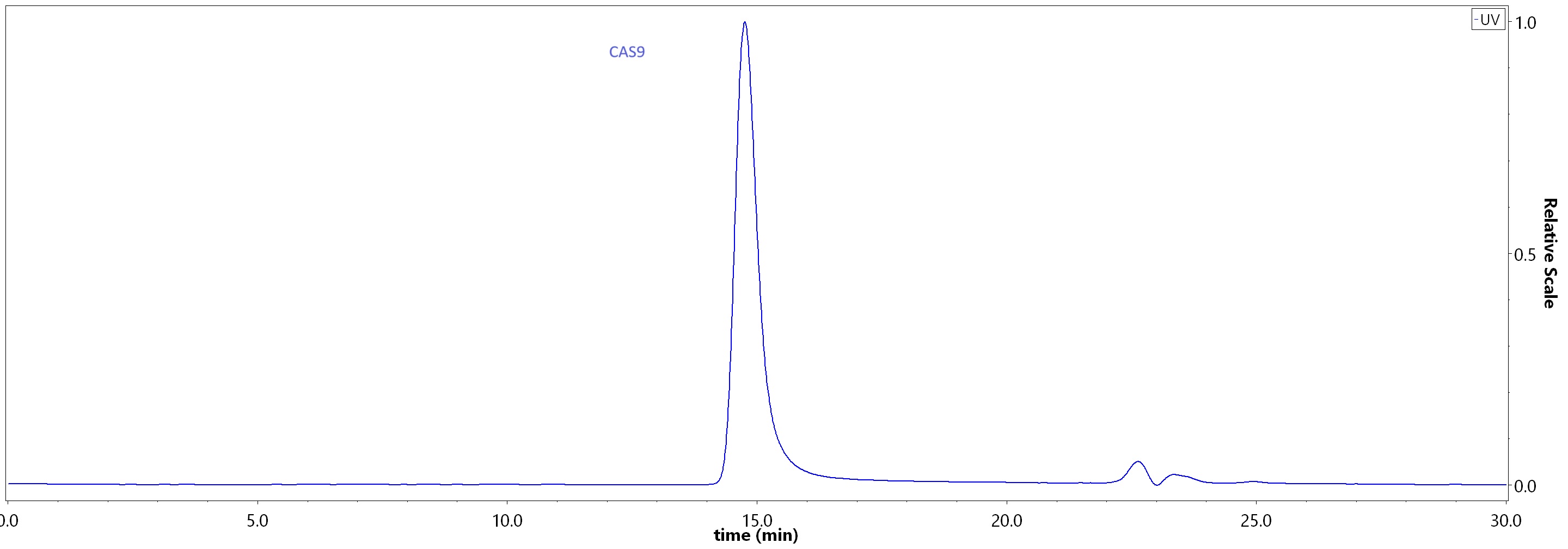
The purity of GMP GENPower™ NLS-Cas9 Nuclease (Cat. No. GMP-CA9S18) was greater than 95% as determined by SEC-HPLC.
活性(Bioactivity)
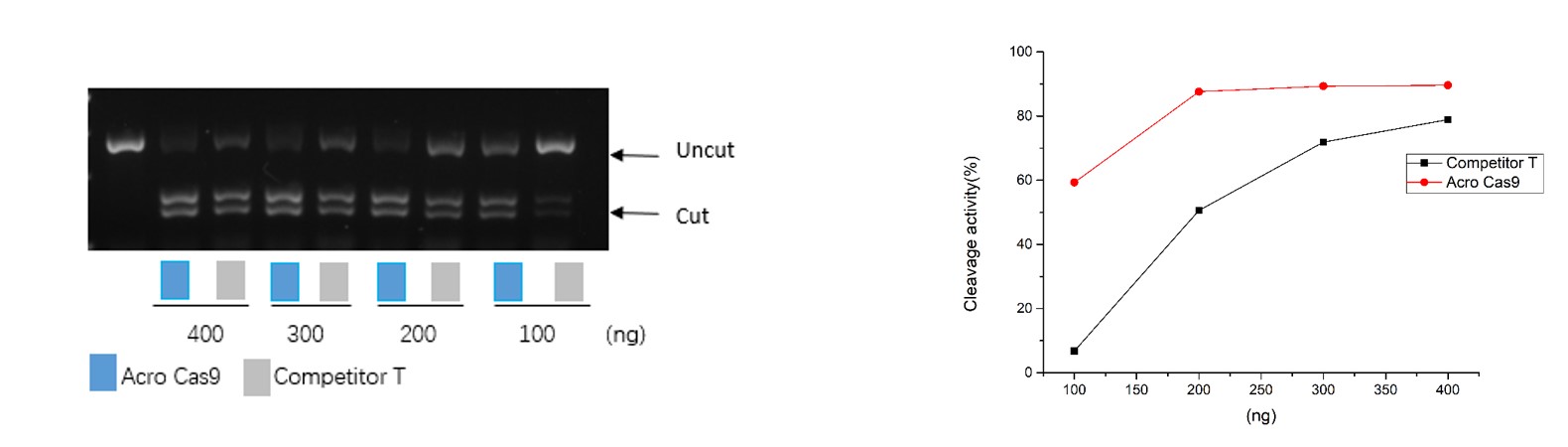
Different amounts of Cas9 were incubated with the same amount of excess gRNA and plasmid for 60 minutes at 37°C. When using 400-200 ng Acro Cas9, the cutting efficiency is greater than 90% (QC tested). In comparison, when using a 200 ng Competitor T, the cutting efficiency is only about 50%.
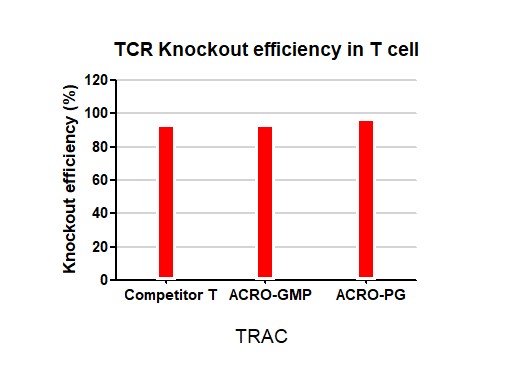
The TCR knockout efficiency with GMP GENPower™ NLS-Cas9 Nuclease in human primary T cells, GMP GENPower™ NLS-Cas9 Nuclease achieved over 95% knockout efficiency.
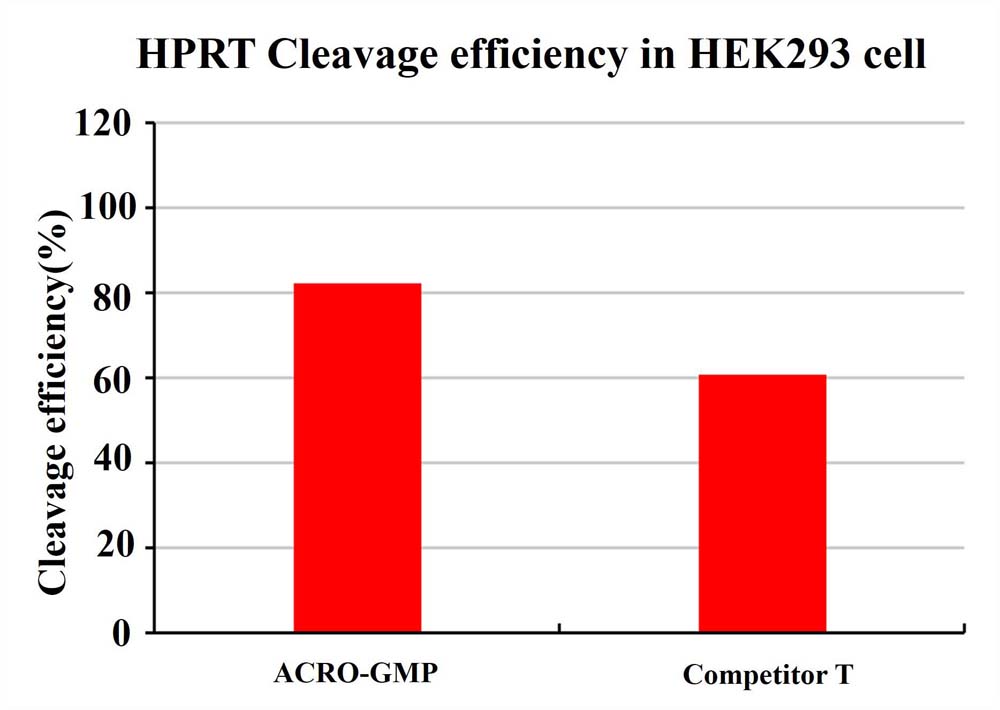
The cleavage efficiency in HEK293 cell 72 hours after electroporation of GMP GENPower™ NLS-Cas9 Nuclease RNP.
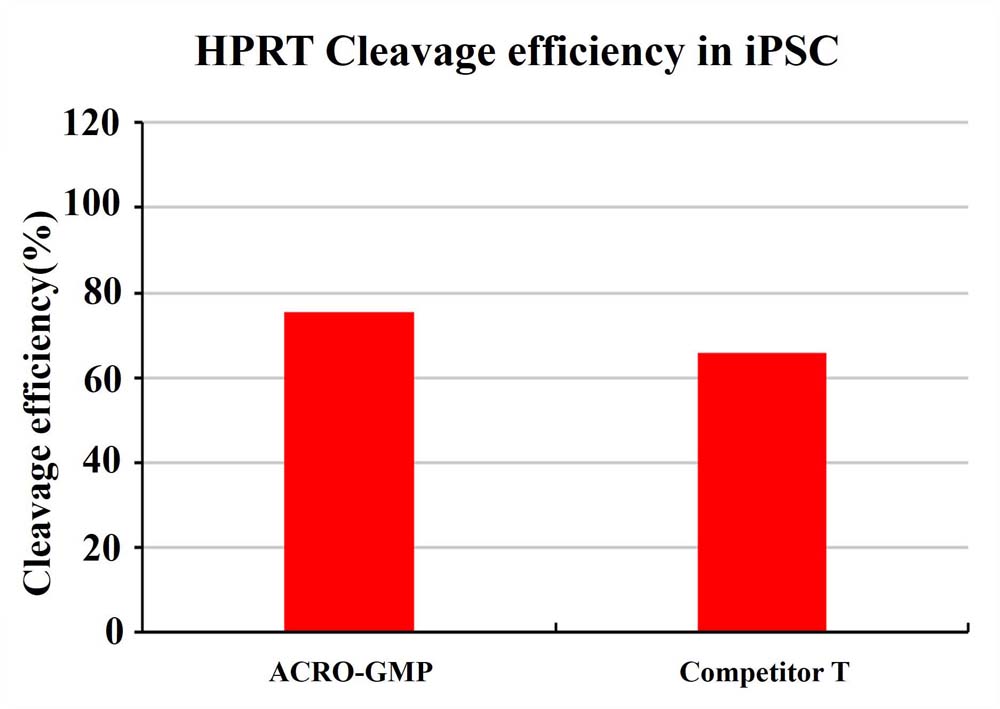
The cleavage efficiency in iPSC 72 hours after electroporation of GMP GENPower™ NLS-Cas9 Nuclease RNP.
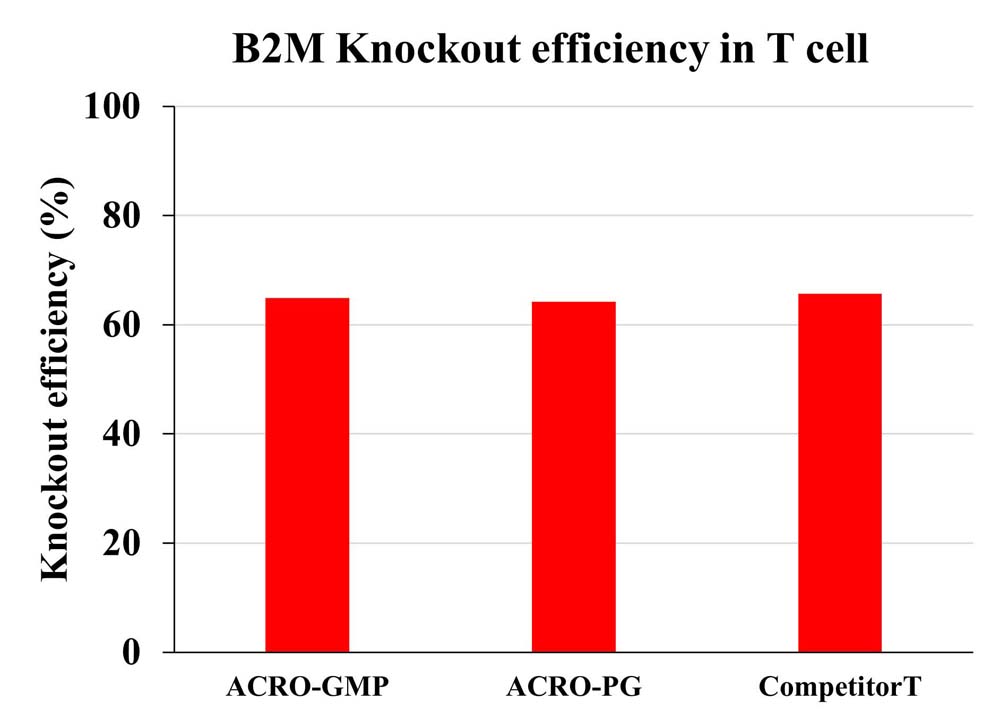
The knockout efficiency for B2M in primary T cell was measured by Flow Cytometry.
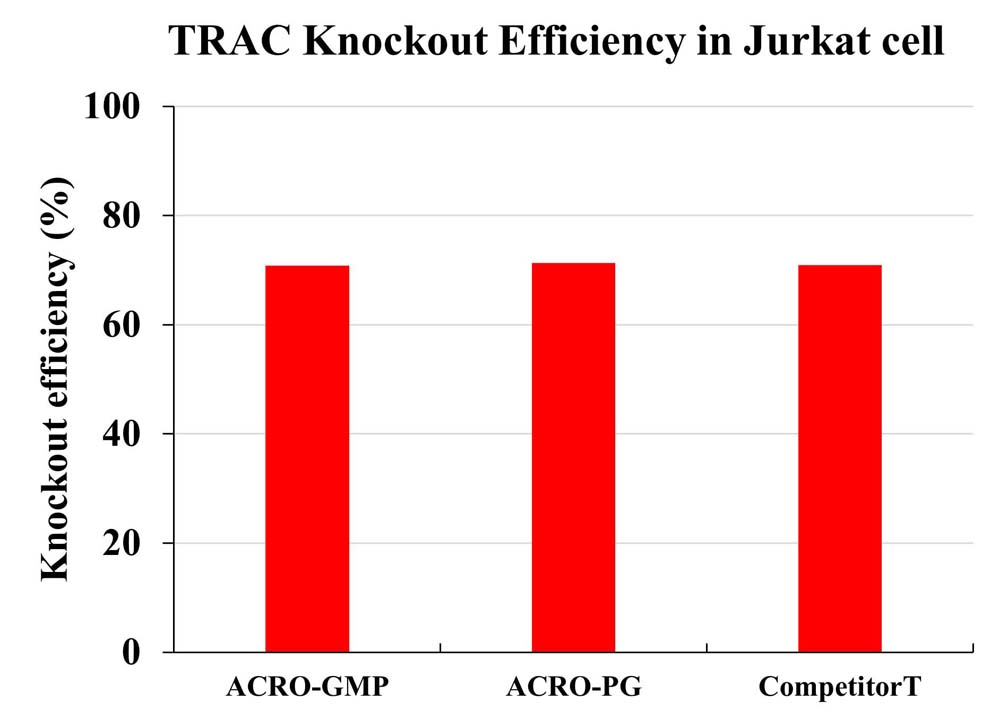
The knockout efficiency for TRAC in Jurkat cell was measured by Flow Cytometry.
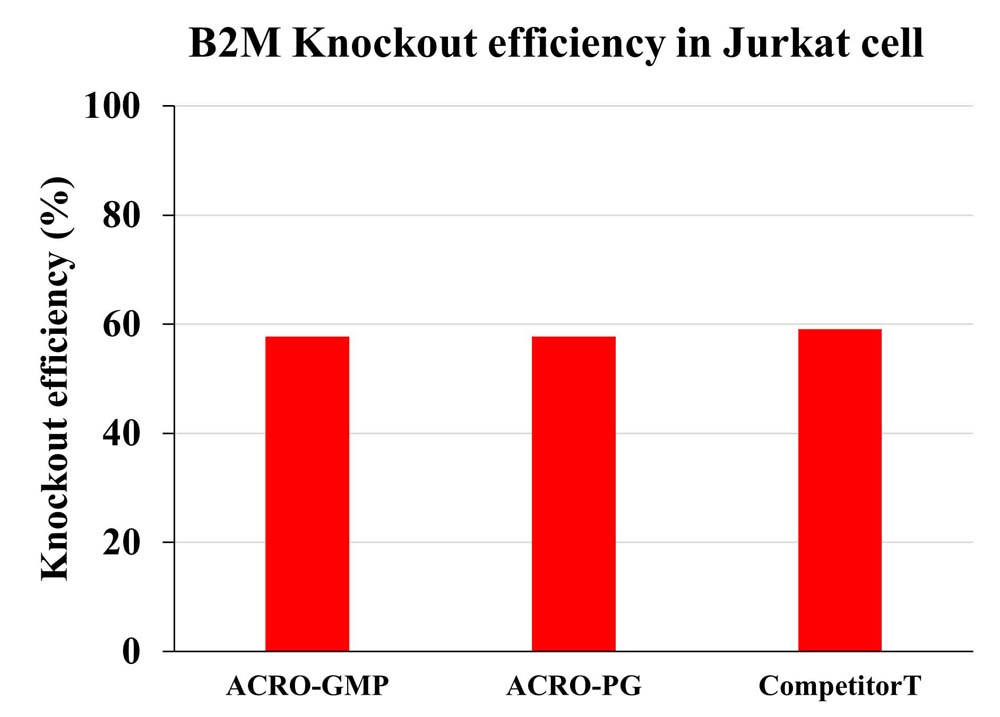
The knockout efficiency for B2M in Jurkat cell was measured by Flow Cytometry.
稳定性(Stability)
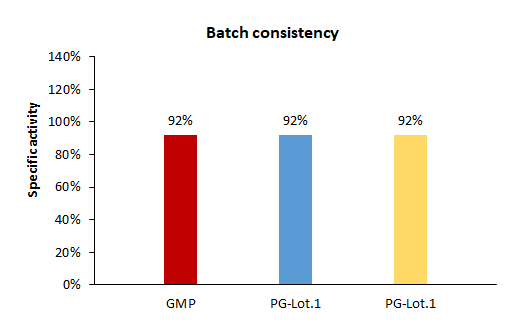
The bioactivity based assay shows batch-to-batch consistency between Acro's GMP and PG Cas9.
用户评价 发表评论

重要声明
MANUFACTURING SPECIFICATIONS
ACROBiosystems GMP grade products are produced under a quality management system and in compliance with relevant guidelines: Ph. Eur General Chapter 5.2.12 Raw materials of biological origin for the production of cell-based and gene therapy medicinal products; USP<92>Growth Factors and Cytokines Used in Cell Therapy Manufacturing; USP<1043>Ancillary Materials for Cell, Gene, and Tissue-Engineered Products; ISO/TS 20399-1:2018, Biotechnology - Ancillary Materials Present During the Production of Cellular Therapeutic Products.
ACROBiosystems Quality Management System Contents:
- Designed under ISO 9001:2015 and ISO 13485:2016, Manufactured and QC tested under a GMP compliance factory.
- Animal-Free materials
- Materials purchased from the approved suppliers by QA
- ISO 5 clean rooms and automatic filling equipment
- Qualified personnel
- Quality-related documents review and approve by QA
- Fully batch production and control records
- Equipment maintenance and calibration
- Validation of analytical procedures
- Stability studies conducted
- Comprehensive regulatory support files
Request For DMFACROBiosystems provide rigorous quality control tests (fully validated equipment, processes and test methods) on our GMP grade products to ensure that they meet stringent standards in terms of purity, safety, activity and inter-batch stability, and each bulk QC lot mainly contains the following specific information:
- SDS-PAGE
- Protein content
- Endotoxin level
- Residual Host Cell DNA content
- Residual Host Cell Protein content
- Biological activity analysis
- Microbial testing
- Mycoplasma testing
- In vitro virus assay
- Batch-to-batch consistency
DISCLAIMER
ACROBiosystems GMP grade products are designed for research, manufacturing use or ex vivo use. CAUTION: Not intended for direct human use.
TERMS AND CONDITIONS
All products are warranted to meet ACROBiosystems Inc.’s (“ACRO”) published specifications when used under normal laboratory conditions.
ACRO DOES NOT MAKE ANY OTHER WARRANTY OR REPRESENTATION WHATSOEVER, WHETHER EXPRESS OR IMPLIED, WITH RESPECT TO ITS PRODUCTS. IN PARTICULAR, ACRO DOES NOT MAKE ANY WARRANTY OF SUITABILITY, NONINFRINGEMENT, MERCHANTABILITY OR FITNESS FOR ANY PARTICULAR PURPOSE.
NOT WITH STANDING ANY OTHER PROVISIONS OF THESE TERMS AND/OR ANY OTHER AGREEMENT BETWEEN ACRO AND PURCHASER FOR THE PURCAHSE OF THE PRODUCTS, ACRO’S TOTAL LIABILITY TO PURCHASER ARISING FROM OR IN RELATION TO THESE TERMS, AN AGREEMENT BETWEEN THE PARTIES OR THE PRODUCTS, WHETHER ARISING IN CONTRACT, TORT OR OTHERWISE SHALL BE LIMITED TO THE TOTAL AMOUNT PAID BY PURCHASER TO ACRO FOR THE RELEVANT PRODUCTS. IN NO EVENT WILL ACRO BE LIABLE FOR THE COST OF PROCUREMENT OF SUBSTITUTE GOODS.
END USER TERMS OF USE OF PRODUCT
The following terms are offered to you upon your acceptance of these End User Terms of Use of Product. By using this product, you indicate your acknowledgment and agreement to these End User Terms of Use of Product. If you do not agree to be bound by and comply with all of the provisions of these End User Terms of Use of Product, you should contact your supplier of the product and make arrangements to return the product.
The End User is aware that ACROBiosystems Inc. and its affiliate (“ACRO”) sell GMP grade products designed for research, manufacturing use or ex vivo use and not intended for human in vivo applications. The End User further agrees, as a condition of the sales of ACRO’s GMP grade products that: a) the End User will not use this GMP grade product in any procedure wherein the product may be directly or indirectly administered to humans, unless the End User has obtained, or prior to their use will have obtained, an Investigational New Drug (IND) exemption from the FDA and will use the product only in accordance with the protocols of such IND and of the Institutional Review Board overseeing the proposed research, or b) the End User will use the products outside of the United States in accordance with the protocols of research approved by the applicable review board or authorized ethics committee and regulatory agencies to which the End User is subject to in their territory.














